Crank Astronomy
(Formerly "Dealing with Creationism in Astronomy")
Creationist Cosmology Issues
- A Changing Speed of Light? (Last Modified: November 24, 2009)
- New Redshift Interpretation (Last Modified: November 24, 2009)
- Polonium Halos (Last Modified: November 24, 2009)
- Accelerated Radioactive Decay? (Last Modified: November 24, 2009)
- Solar Claims (Last Modified: November 24, 2009)
- A Shortage of Supernova Remnants? (Last Modified: November 24, 2009)
- A Decaying Magnetic Field of the Earth (Last Modified: November 24, 2009)
- Redshift Quantization (Last Modified: March 18, 2012)
- "Relativistic" Creationist Cosmology (Last Modified: March 18, 2012)
- Anomaly Science (Last Modified: November 24, 2009)
- Development Notes (Last Modified: November 24, 2009)
Site Resources
- History & Technology (Last Modified: March 18, 2012)
- Philosophy & Religion (Last Modified: November 24, 2009)
- Classroom Resources (Last Modified: November 24, 2009)
- Presentation Slides (Last Modified: November 24, 2009)
Credits
Site Contents
© 2001-2012, W.T. Bridgman
Send comments, questions, and other inquiries to
This site is also listed at comPADRE and AstronomyCenter.org.
CSS developed with the assistance of CSScreator
Claims of Accelerated Radioactive Decay
Here we explore some simple implications of the poorly done science supporting the idea of extensive accelerated radioactive decay, specifically those advocated by the ICR in the RATE ("Radioactivity and the Age of The Earth") document and at Answers in Genesis. Accelerated radioactive decay is occasionally invoked by creationists to explain radiometric dating results. By retreating to this position, they are essentially abandoning claims that isochron dating is seriously flawed. Here we examine some simple requirements for accelerated decay to effectively "mimic" the appearance of long ratioisotopic ages and some of the problems with currently proposed mechanisms for accelerating decay rates.
Here we consider two separate sets of claims
- Claims of Accelerated Radioactive Decay in "Radioactivity and the Age of The Earth" (RATE)
- Accelerated Radioactive Decay According to Answers in Genesis
There are a number of online resources on the principles underlying radioactive decay.
- See Wikipedia: Isochron dating
- See Wikipedia: Age of the Earth
- See Wikipedia: Radiometric dating
- See TalkOrigins.org: Isochron Dating. This is a very good explanation of isochron dating demonstrating how the method checks against many types of errors.
Claims of Accelerated Radioactive Decay in
"Radioactivity
and the Age of The Earth" (RATE)
- Radioactivity and the Age of The Earth AKA ICR Impact #301. (Is this Volume I? There seems to be a Volume II advertised in the ICR store.)
- Helium Evidence for A Young World Remains Crystal-Clear by D. Russell Humphreys
Rebuttal Links:The articles linked below deal with helium diffusion claims and geology used in RATE.
- "RATE" Leaders Abandon Geologic Fantasies and Admit that Extensive Radioactive Decay has Occurred by Kevin R. Henke, Ph.D.
- Young-Earth Creationist Helium Diffusion "Dates": Fallacies Based on Bad Assumptions and Questionable Data by Kevin R. Henke, Ph.D.
Problems with Accelerated Radioactive Decay
Page numbers correspond to the RATE document linked above.
- In DeYoung's discussion of how accelerated decay can solve the age issue, he presents a simple example (pg 42). An important point in this mechanism is that it requires that all isotopes have their decay rates accelerated by the exact same numerical factor, for all types of decay if it is to mimic the behavior of assumed constant decay rates.
- Using Humphrey's model for accelerated radioactive decay (pg 357-364), we can show that for a given difference in pion mass, the change in alpha-decay rate will not be the same for all elements or all isotopes of the same elements.
- Humphreys model for accelerated beta-decay has similar problems (pg 364). The explicit dependence on atomic number means the acceleration factor will be different for each isotope. The additional dependence on the mass of the W-boson complicates the analysis only slightly.
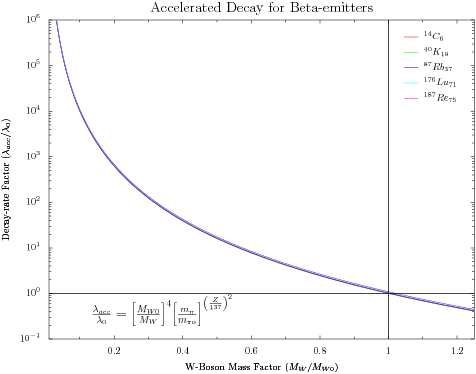
Here we combine the pion mass determined above and look for a W-boson mass change to accelerate the decay of Re-187 by the same factor of 750,000. We find a W-boson mass factor of 0.034724 which alters the decay rates of other beta-emitters by significant amounts (though not a dramatic as the pion mass changes). Unfortunately, the scaling makes it difficult to distinguish the different elements. Computed values in the table below. Beta Emitter Acceleration Factor C-14 6.88e+05 K-40 6.92e+05 Rb-87 7.02e+05 Lu-176 7.43e+05 Re-187 7.50e+05 - Humphreys' mechanism changes the fundamental forces binding nuclei together. In making existing unstable nuclei less stable, it opens the possibility that the mechanism will also make currently stable nuclei ustable. This creates additional problems for atoms in living tissue beyond the C-14 and K-40 isotopes Humphreys discusses on page 373.
- Humphreys derived Equations 8 (pg 362) and 12 (pg 364) for rate changes from formulae that were already based on some approximations. With such significant changes in decay rates requiring large changes in pion and W-boson masses, a re-examination of the input assumptions in their derivations would be in order.
- While they mention there may be some cosmic events based on accelerated decay (pg 374), they ignore the fact that there are existing cosmological observations (Index of Creationist Claims, Claim CF210) attesting to the constancy of these rates in the distant past.
The interesting thing about this analysis is that these flaws in Humphreys model were immediately apparent. I identified them on my first reading of the RATE document. The fact that Z and/or A appeared in the rate equations was a dead giveaway that these rates would vary by element and isotope. It took me an evening to estimate these values, an additional few days to explore other implications and generate nice graphics.
These errors were trivial to find. They could be recognized by a bright high-school student, yet none of the Ph.D.s at the ICR, including Humphreys, noticed before they made it into print. This points to a very serious failure in their peer-review process (though it is far from the first...).
Accelerated Radioactive Decay According to
Answers in Genesis
At Answers in Genesis (hereafter referred to as AIG) their "model" for decay acceleration is non-existent beyond promoting the experimental work of others as supporting their claims. AIG has propagated some misinformation about recent experiments which suggest radioactive decay rates can vary significantly.
- AIG: Radioactive decay rate depends on chemical environment by Tas Walker
- AIG: Billion-fold acceleration of radioactivity demonstrated in laboratory by John Woodmorappe
What is it that AIG doesn't tell you?
- The issue mentioned in the RATE claim, that all rates must vary by the exact same factor (to preserve the currently observed agreement between different dating methods), is not enforced. Screening mechanisms will alter the rates of different elements by differnt factors.
- Electron screening altering nuclear reaction rates is an issue in astrophysical environments known for a number of years (and in some cases, decades).
- The experiments are basically confirming theoretical calculations for astrophysical environments, which further demonstrates that mathematical extrapolations of these physical princples is valid.
- The physical environment (temperature and electron density) necessary to achieve significant changes in decay rates for elements using these mechanisms in a geological context would reset the radiometric clock. More accurately, they would destroy the geological environment necessary to perform isochron dating.
The authors of the original paper (Dependence of the decay rate of 7Be on chemical forms) make the statement
"The observed difference, by as much as 1.5%, clearly indicates that decay rates of nuclides undergoing electron capture decay are not necessarily constant as has always been assumed in geological, oceanographic, and environmental studies."
The authors seem oblivious to the history of this reaction. That the beryllium-7 decay rate could be influenced by the surrounding electron environment was explored for astrophysical environments by John Bahcall as far back as 1962 (Electron Capture and Nuclear Matrix Elements of Be-7). For Bahcall, this issue was important in the proton-proton chain powering the Sun and other stars as well as understanding the issues of neutrino production. The experiment approximates the electron density of the astrophysical environment in a laboratory setting and successfully demonstrated that electron screening can alter decay rates under these conditions, as predicted over forty years earlier.
In other articles, such as Radioactivity speeds up (September 2004), it's made much clearer that these results are primarily applicable to extreme astrophysical environments:
"Increasing the decay rate by just 0.83% will have little effect on radioisotopes with half-lives of thousands or millions of years. However, Ohtsuki and co-workers say that their results will help identify the sort of environments that increase the decay rate. These could include the high-pressure conditions found inside neutron stars."
While I've yet to find an article at AIG or other creationist organization on this result (A cool solution to waste disposal (July 2006)), I'll include an evaluation here. Here again, we see that different decays will occur at different rates:
"But Rolfs realized that the reverse reaction might also occur and that free electrons could enhance the ejection of positively charged particles from a nucleus. This would reduce the half-lives of alpha-decay or beta+-decay, and increase half-lives for processes involving electrons (which are repelled by the free electrons within the metal), i.e. beta--decay and electron capture."
In the original article (High-Z electron screening: the cases 50V(p,n)50Cr and 176Lu(p,n)176Hf), the authors explain their analysis. I see several problems with their attempts to extrapolate the accelerated decay rates to factors of hundreds and more:
- The extrapolation of the Debye screening mechanism down to 4K is the main problem. They are apparently using the plasma approximation for deriving the Debye radius and plasma parameter. I'm still tracking down some details on this but it appears the approximation used in this screening, while valid in the range of the experiment, would not be valid at 4K where they claim large changes in decay rates.
- You have a block of radioactive material with an accelerated rate of decay (and energy output). How are you goint to keep it at cryrogenic temperatures?
Last Modified: Tue Nov 24 23:41:17 2009
